About the Prize
The Brucellosis Challenge
Brucellosis is a costly disease that affects ruminants (cattle, sheep, goats, buffalo, etc.) and causes abortions, infertility, decreased milk production, and weight loss, amongst other effects. Brucellosis is endemic in a number of developing countries, and the toll on smallholder farmers (SHFs) in South Asia and Sub-Saharan Africa is particularly devastating, since cattle and small ruminants, such as goats and sheep, are a crucial source of income for this population. The annual impact of Brucellosis to SHFs across Sub-Saharan Africa and South Asia is estimated at USD $501 million per year.
Brucellosis is also the most common type of disease that is transmitted from animals to humans, with approximately 500,000 new human cases reported each year.
Wholesale vaccination of livestock can be a cost-effective way of controlling the disease and limiting its impact on both human and animal health. However, the existing Brucellosis vaccines are not safe or efficacious enough to use in developing countries. Current vaccines do not provide protection across different species of animal hosts, are unsafe for use in pregnant animals, have variable efficacy, and can harm humans. A new vaccine that addresses these shortcomings would deliver lasting benefits to SHFs’ health and livelihoods.
What are we looking for?
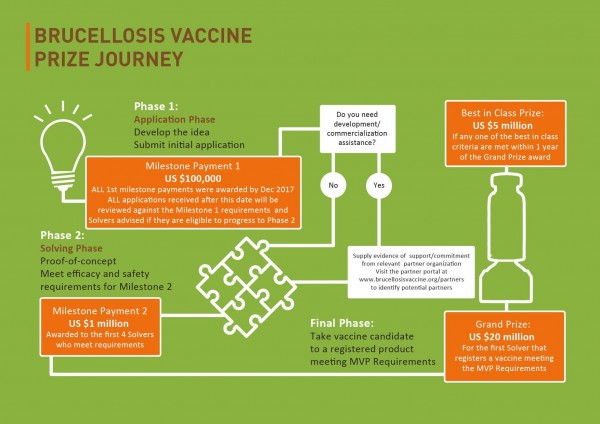
The Brucellosis Vaccine Prize is a global US $30 million competition funded by AgResults (a collaborative initiative between the governments of Australia, Canada, the UK and the US as well as the Bill & Melinda Gates Foundation), and implemented by the Global Alliance for Livestock Veterinary Medicines (GALVmed).
AgResults and GALVmed are looking for animal health innovators to develop and register a vaccine that is safe, efficacious, and viable for use against Brucella melitensis in small ruminants across the developing world.
We support collaboration between animal health innovators in achieving this aim and encourage organisations to use the Partnering Page to identify potential partners.
The winning vaccine will need to meet all the requirements set out in the Minimum Viable Product (MVP), and will ideally overcome all current hurdles that inhibit the safety and efficacy of current B. melitensis vaccines in developing countries.
Considerations for the MVP include:
- Species: B.melitensis in sheep or goat
- Safe: Safe for pregnant animals (Less than 5% abortion)
- Efficacious: More than 80% animals protected
- Convenient: Single vaccination annually
- Smallholder Farmer Friendly: Affordable cost, long shelf life
Full details on the Minimum Viable Product (MVP) requirements can be found in Appendix 3 of the official Competition Rules.
(Archived Competition Rules: January 2020)
(Archived Competition Rules: April 2018)
(Archived Competition Rules: November 2016)
For the Grand Prize of US $20 million, the winning vaccine will need to both meet the requirements of the MVP and obtain regulatory registration in any one of the following regions/countries: EU, Japan, USA, Canada, or Australia.
Full details on vaccine registration requirements can be found in Section 7 of the official Competition Rules.
(Archived Competition Rules: January 2020)
(Archived Competition Rules: April 2018)
(Archived Competition Rules: November 2016)
Best in Class
An additional US $5 million prize will be available to Solvers that develop a vaccine that meets the MVP requirements and any one of the Best in Class criteria. The Best in Class prize will be open to Solvers for one year after the award of the Grand Prize.
Best in Class criteria include:
- Cross - Species Protection: Protect against both B. melintensis in small ruminants and B.abortus in cattle
- Safety: Providing maximum human and animal safety (e.g. inactivated)
- Thermoresistance: Effective at 45 degrees centigrade for 3 weeks
- Curative: Curative/therapeutic effect on infected animals
Full details on the Best in Class requirements can be found in the in Appendix 4 of the official Competition Rules.
(Archived Competition Rules: January 2020)
(Archived Competition Rules: April 2018)
Prize Structure and Milestone Phases